CEO Simon Bunyatov
Sterilization Suite Design & Construction
OSHA, CDC, and infection-control-compliant sterilization centers — designed for proper workflow and built to inspect-clean.
About Sterilization Suite Design & Construction
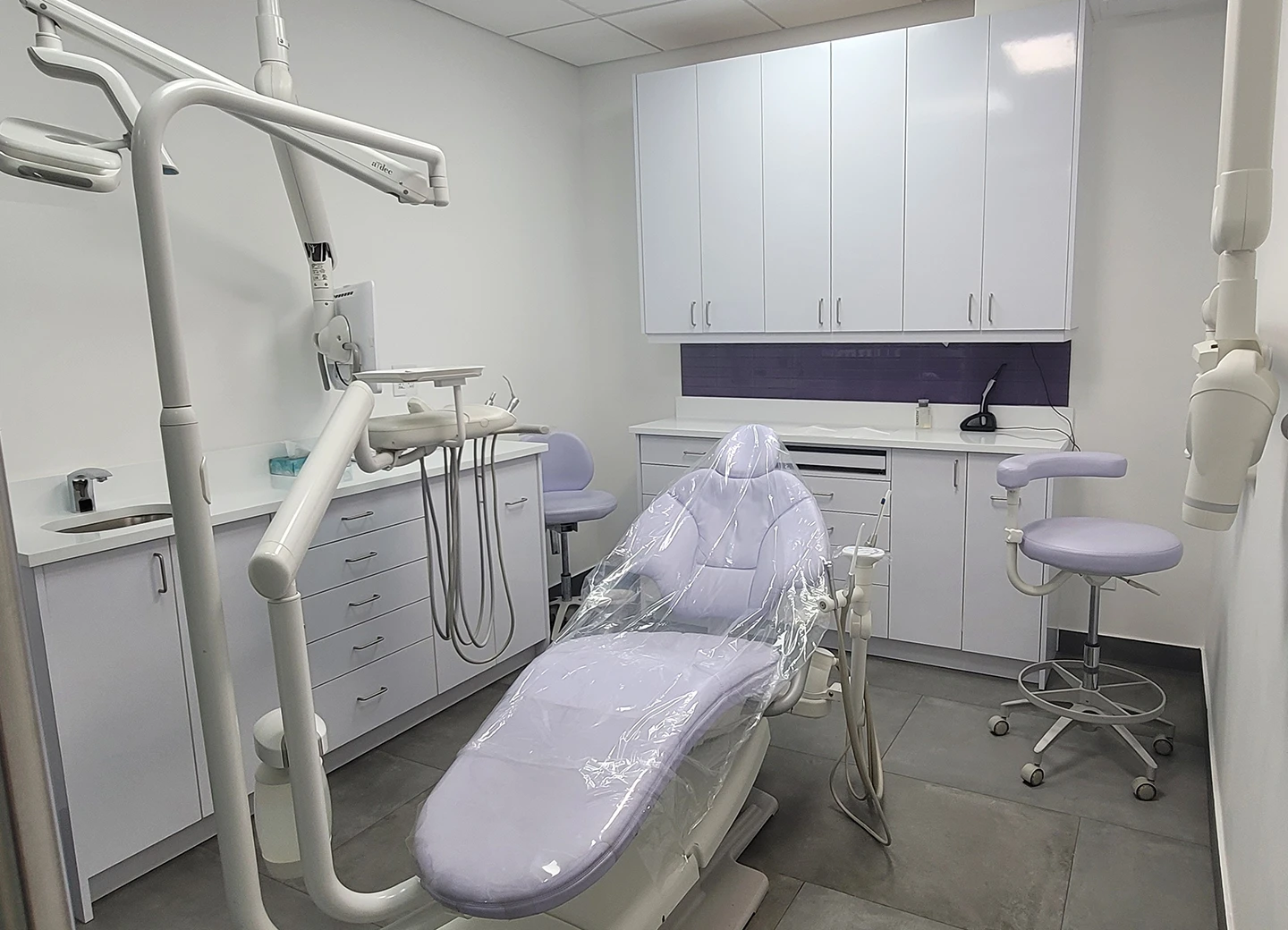
The sterilization suite is the operational heart of any dental or medical practice. Workflow problems here cause infection-control failures, inspection issues, and slow turn times. DBF Studio designs and builds sterilization suites with proper dirty-to-clean directional flow, appropriate ventilation, code-compliant finishes, and equipment integration that meets OSHA, CDC, and current best-practice infection-control standards.
Why DBF Studio
We design every sterilization suite around the actual instrument workflow — receiving, decon, packaging, sterilization, storage — with one-way directional flow that prevents cross-contamination. Stainless steel tops, sealed wall finishes, and hands-free fixtures specified to standard. Equipment integration coordinated for major sterilizer and decon manufacturers.
Scope of Work
Sterilization suite components
- Receiving and decontamination zone (dirty side)
- Cleaning and packaging zone
- Sterilization zone (autoclave bank)
- Sterile storage zone (clean side)
- Distribution to operatories
Workflow & compliance
- One-way dirty-to-clean directional flow
- OSHA bloodborne pathogen compliance
- CDC infection-control guideline adherence
- Hands-free sinks, eyewash stations, and PPE access
- Documented air changes and ventilation
Finishes & utilities
- Sealed, scrubbable wall and floor finishes
- Stainless steel countertops and backsplashes
- Plumbing for autoclaves, ultrasonic cleaners, and rinse stations
- HVAC zoning with appropriate filtration
- Dedicated electrical for sterilization equipment
Equipment integration
- Autoclave installation coordination
- Ultrasonic and instrument washer integration
- Custom millwork for sterilization workflow
Our Process
- Workflow analysis. We map your instrument volume, peak sterilization load, and current bottlenecks before designing the room.
- Layout & equipment plan. Architectural drawings, equipment layout, and utility plan.
- Construction. Most standalone sterilization suite builds run 3–6 weeks.
- Equipment install. Manufacturer techs coordinated for autoclave, ultrasonic, and washer install.
- Walkthrough & documentation. Final inspection plus a workflow documentation package for your infection-control program.
Selected Projects
A few representative projects from our portfolio.
Cost & Timeline
Standalone sterilization suite construction typically runs $25,000–$75,000 depending on size, equipment scope, and finish level. Suites built as part of a larger practice buildout are scoped within the total project.
Permits & Compliance
Sterilization suite renovations typically file Alt-2. We handle DOB filings and inspections. The suite as-built will pass OSHA, CDC, and current infection-control program audits as a baseline.
“He addressed every concern with professionalism while respecting our budget requirements. We are excited to move forward with another project with DBF Studio.”
Frequently Asked Questions
How big should my sterilization suite be?
Most general dentistry practices run 80–150 sq ft of dedicated sterilization. Multi-specialty and high-volume practices may need 200–300 sq ft. We size against your instrument volume and peak sterilization load.
What’s the dirty-to-clean workflow standard?
Instruments enter at the dirty side (receiving and decon), move through cleaning and packaging, then sterilization, then to a separate clean storage zone before distribution to operatories. The room is laid out so cross-contamination is physically difficult.
What finishes are required?
Sealed, scrubbable wall and floor surfaces. Stainless steel countertops at decon and sterilization zones. Hands-free fixtures. Coved base flooring is recommended.
Will my suite pass OSHA inspection?
Yes. Every suite we build is designed to current OSHA bloodborne pathogen and CDC infection-control standards. We document workflow and finishes for your records.
Do you coordinate autoclave install?
Yes — we coordinate with autoclave, ultrasonic, and washer manufacturers’ certified techs for install and acceptance testing.
How long does a sterilization suite take to build?
Three to six weeks for a standalone build into existing finished space. Often run nights and weekends to avoid disrupting practice operations.
Can you upgrade my existing suite without a full rebuild?
Often yes. Workflow corrections, finish upgrades, and equipment additions can be staged. We assess existing conditions and propose targeted upgrades.
How do we start?
Call (917) 681-4821 or send a consultation request. We’ll walk your existing suite, assess workflow, and produce a written proposal.